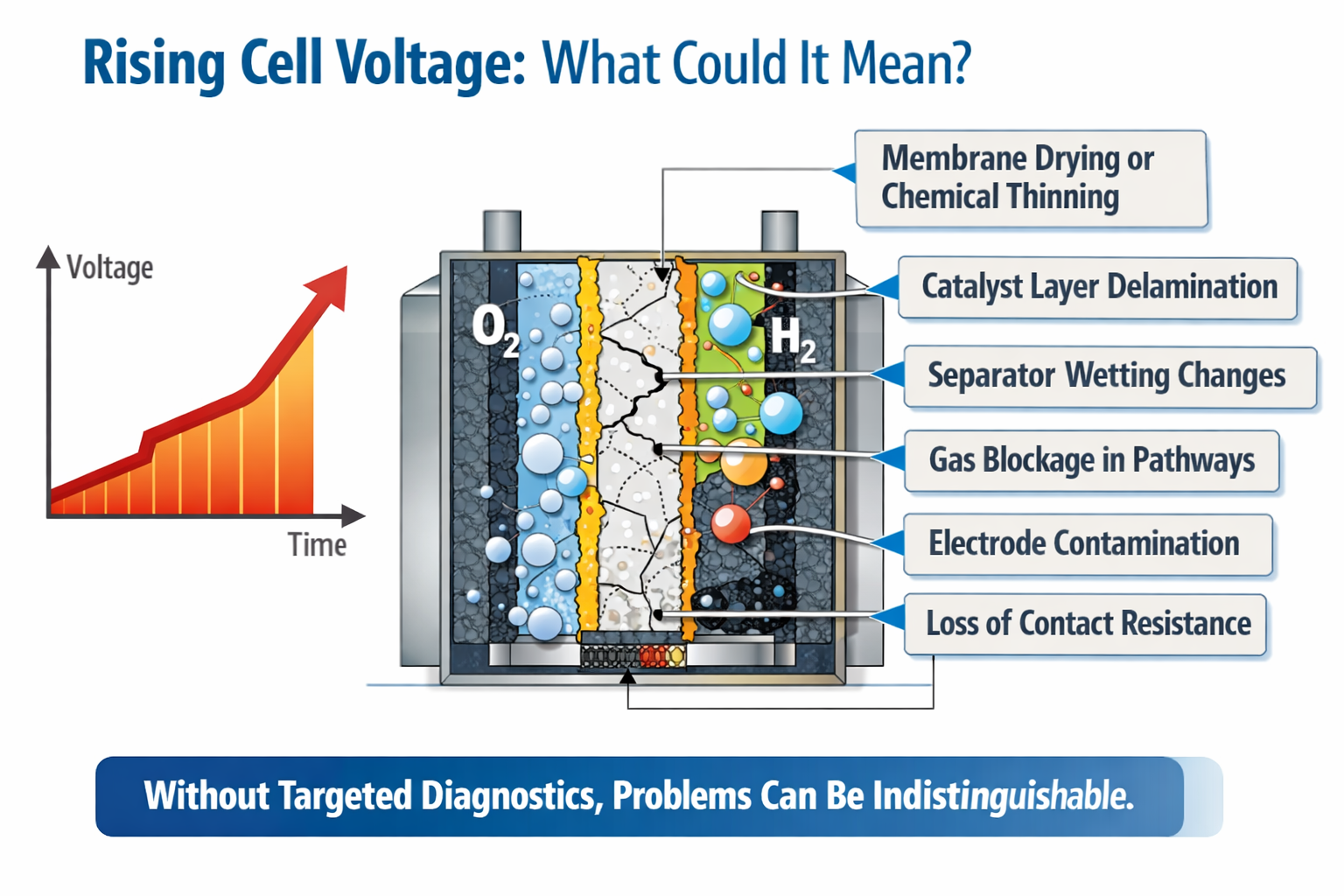
The key point is simple: performance loss is rarely a single failure event. It’s a collection of small, interacting changes,chemical, mechanical, and transport-related, that accumulate until voltage rises, efficiency drops, gas purity suffers, and maintenance becomes inevitable. That is why monitoring is not an academic luxury. It is the shortest path to understanding which mechanism dominates, where it happens, and how to stop it.
That is why I believe the most powerful question in hydrogen R&D is not “how high can we push the current density?” but rather: “What happens after 1,000… 10,000… 30,000 hours, and how do we know early enough to do something about it?”
Alkaline and Anion exchange electrolysis: the cost promise comes with a durability bill
At CIC energiGUNE, we place a strong emphasis on alkaline water electrolysis (AWE) and anion-exchange membrane water electrolysis (AEMEL), because these platforms offer a compelling cost pathway: non-precious catalysts, less dependency on critical raw materials, and potentially simpler stack architectures.
But the price advantage only becomes real if durability follows.
AEM electrolysis, in particular, sits in an interesting “middle ground”: it wants to combine the compactness and differential-pressure operation of PEM (Proton Exchange Membrane)-style MEAs (Membrane Electrode Assembly) with the catalyst flexibility of alkaline chemistry. The challenge is that AEM systems concentrate a lot of complexity in the catalyst layer and ionomer: the ionomer must bind catalyst particles, provide OH⁻ conductivity pathways, and survive strongly alkaline and oxidative conditions, especially near the anode. Recent literature is increasingly explicit that the ionomer in the catalyst layer can be the limiting component, not just the membrane itself.
And then there is classic alkaline electrolysis, where the separator/diaphragm and the two-phase flow environment are decisive. Alkaline systems are often seen as “mature,” but modern AWE is evolving quickly toward zero-gap and narrow-gap designs, higher current densities, and thinner separators, all of which raise the stakes for controlled gas removal, crossover management, and mechanical robustness.
What actually degrades? Three families of failure modes you can’t ignore
-
The ion-conducting barrier (membrane, diaphragm, separator)
This component has one job: allow ions through while keeping gases apart. In the real world it also has to survive pressure gradients, temperature swings, chemical attack, swelling/shrinkage cycles, and impurity exposure.
In alkaline and AEM systems, a recurring trade-off appears again and again:
- Thinner = lower ohmic losses, but higher vulnerability to pinholes, mechanical creep, and crossover risk.
- Smaller pores / higher tortuosity = lower crossover, but higher resistance and more sensitivity to wetting and bubble management.
In other words, separator engineering is not “passive”: it’s a central design lever, and a common failure origin.
-
The catalyst layer and its interfaces (where performance is born and where it can quietly die)
In AEM electrolysis, durability is often determined within the catalyst layer, including the catalyst, ionomer, pore structure, and transport layers. Even if the catalyst is stable, the system can degrade if the ionomer loses conductivity, detaches from the catalyst surface, or chemically fragments under oxidative stress.
For PEM, the story is different but equally instructive: expensive iridium-based anodes remain the benchmark, yet operando studies show that catalysts restructure under oxygen evolution, forming highly disordered active oxide motifs that evolve with potential and current. This matters because it reminds us of a universal principle: the active state is often not the as-manufactured state, and degradation sometimes starts as “normal activation” that slowly drifts too far.
-
Two-phase transport (bubbles are not just a nuisance; they are a degradation driver)
This is where alkaline and AEM really demand attention. Gas evolution creates bubbles everywhere, inside porous electrodes, at catalyst surfaces, in channels, and within small gaps. If you manage bubbles well, you improve efficiency. If you manage them poorly, you create local dry-out/flooding, current hotspots, and uneven aging.
The takeaway from several studies is blunt: bubble physics is lifetime physics. Two-phase flow determines local resistances, local concentrations, and local mechanical stress, and those local conditions decide which component fails first.
|
Failure Domain |
What Typically Fails |
PEM |
AEM |
Alkaline |
|
Ion-Conducting Barrier |
Conductivity, gas separation |
Membrane thinning, radical attack |
Stability–conductivity trade-off, crossover |
Diapagm aging, carbonate effects |
|
Catalyst Layer & Interfaces |
Catalyst–ionomer stability |
Ir restructuring and dissolution under OER |
Ionomer chemical degradation dominant |
Ni corrosion, interface detachment |
|
Two-Phase Transport |
Bubble-induced heterogeneity |
Moderate sensitivity |
High sensitivity |
Very high sensitivity |
Why monitoring changes the game: from post-mortem to prevention
Traditional durability work often follows a painful script: run for hundreds of hours, observe a voltage rise, disassemble the cell, and try to guess the cause. The problem is that many degradation pathways leave similar electrical fingerprints.